Space-filling model of parent peptide V13K L . Hydrophobic amino acids... | Download Scientific Diagram

Binding of cationic peptides (KX)4K to DPPG bilayers. Increasing the hydrophobicity of the uncharged amino acid X drives formation of membrane bound β-sheets: A DSC and FT-IR study - ScienceDirect

Interactions of Cationic-Hydrophobic Peptides with Lipid Bilayers: A Monte Carlo Simulation Method: Biophysical Journal

Characterizing hydrophobicity of amino acid side chains in a protein environment via measuring contact angle of a water nanodroplet on planar peptide network | PNAS

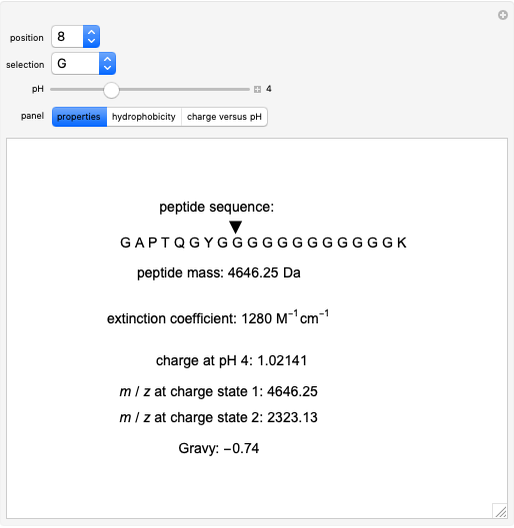
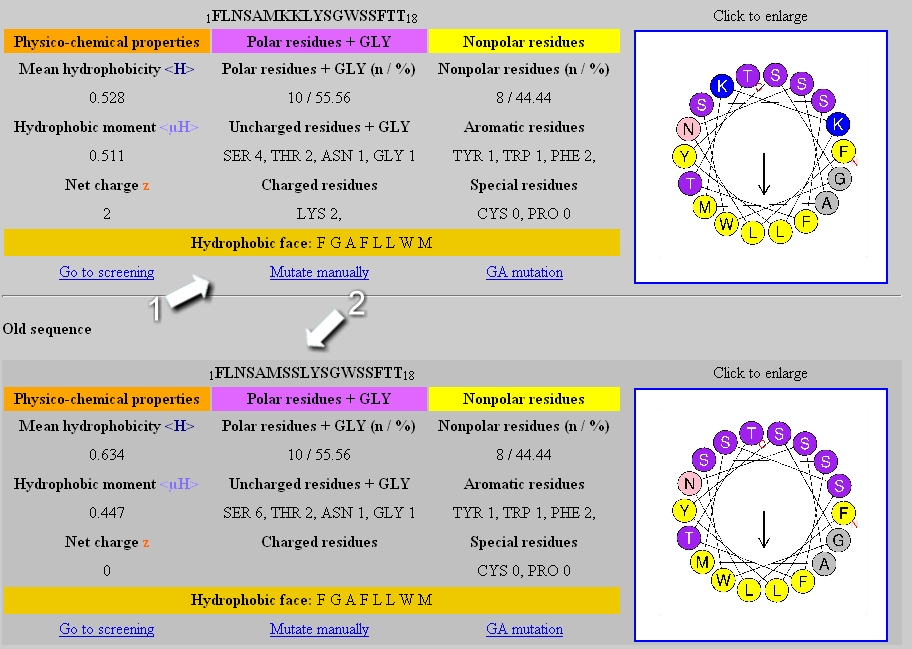
Mean hydrophobicity, hydrophobic moment, net charge and helical wheel... | Download Scientific Diagram
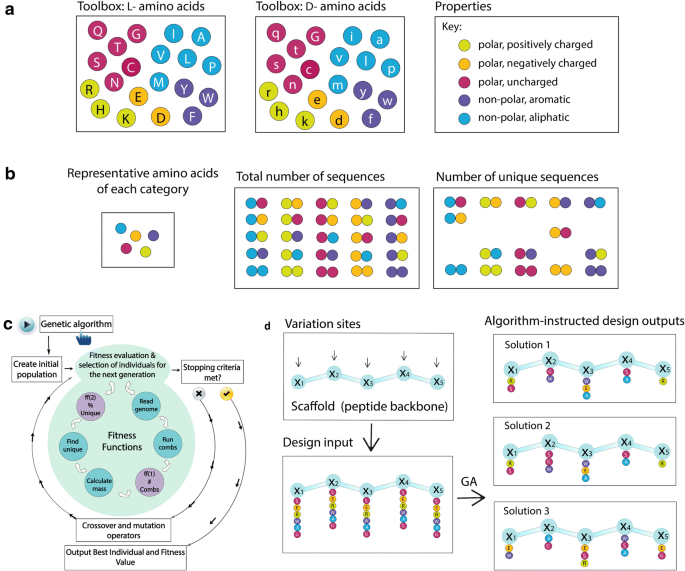
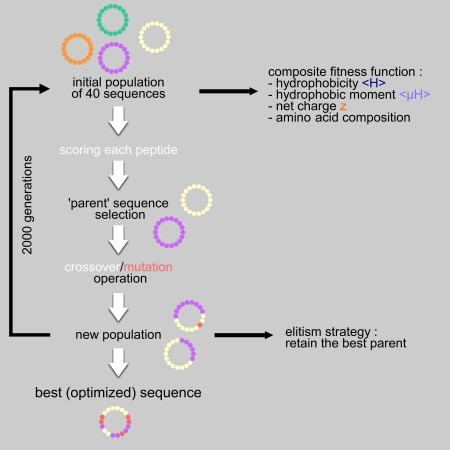
Algorithm-supported, mass and sequence diversity-oriented random peptide library design | Journal of Cheminformatics | Full Text

Characterizing hydrophobicity of amino acid side chains in a protein environment via measuring contact angle of a water nanodroplet on planar peptide network | PNAS
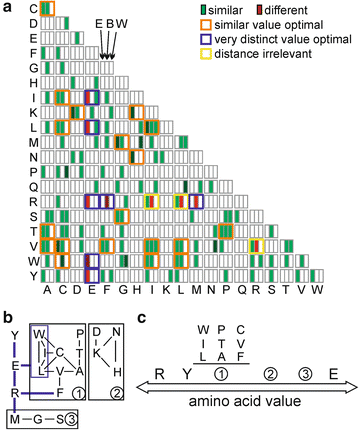
50 years of amino acid hydrophobicity scales: revisiting the capacity for peptide classification | Biological Research | Full Text

Efficient transdermal delivery of functional protein cargoes by a hydrophobic peptide MTD 1067 | Scientific Reports

Clustering of Large Hydrophobes in the Hydrophobic Core of Two-stranded α-Helical Coiled-Coils Controls Protein Folding and Stability* - Journal of Biological Chemistry
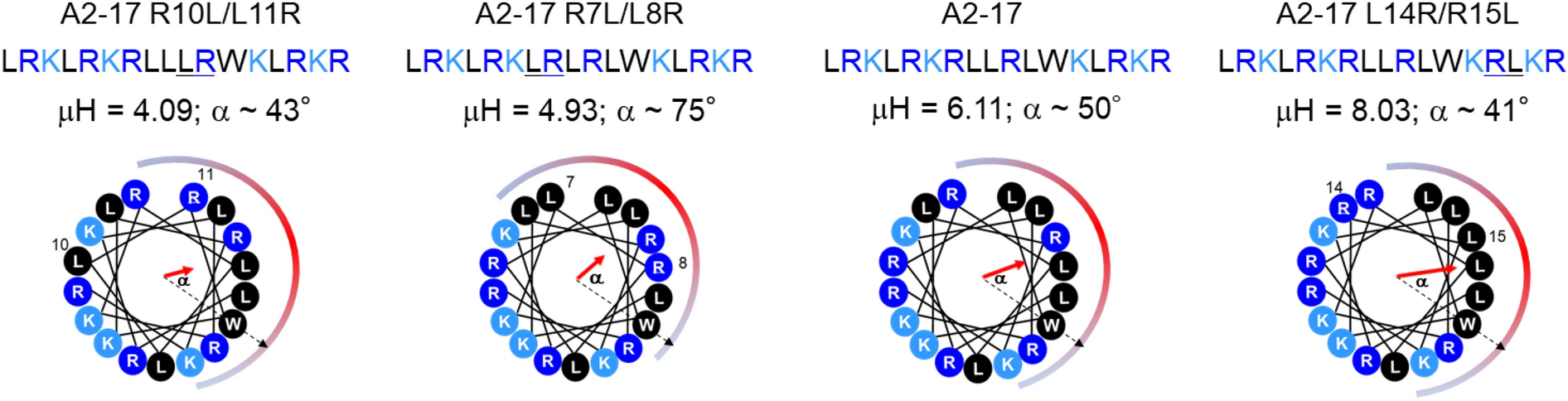
Effect of hydrophobic moment on membrane interaction and cell penetration of apolipoprotein E-derived arginine-rich amphipathic α-helical peptides | Scientific Reports

Solubility test of NSP and NSP r. (A) Peptide models computed by the... | Download Scientific Diagram

Equation 1 was used to determine effective hydrophobicity. The helical... | Download Scientific Diagram

Analysis of hydrophobic and hydrophilic moments of short penetrating peptides for enhancing mitochondrial localization: prediction and validation - Pirisinu - 2019 - The FASEB Journal - Wiley Online Library